Neurodegenerative diseases are typically composed of syndromes with similar clinical features encompassing differing etiologies and heterogeneous responses to treatment. The development of novel disease modifying treatment is often hindered by long and expensive Phase II studies and relies on the outcomes to progress the novel treatment into later phases.There is an urgent need for sensitive molecular markers of neurodegenerative disease progression that provide proof of principle that a novel therapeutic affects the underlying pathophysiology. These molecular markers would allow for small, targeted trials of novel therapies providing readouts in smaller cohorts and in shorter time frames, enabling the selection of promising new treatments for pivotal Phase III trials. The MIND MAPS program was developed to address these needs and is a collaboration, bringing together top-tier academia and industry with the aim of developing imaging biomarkers of the neurodegenerative process.
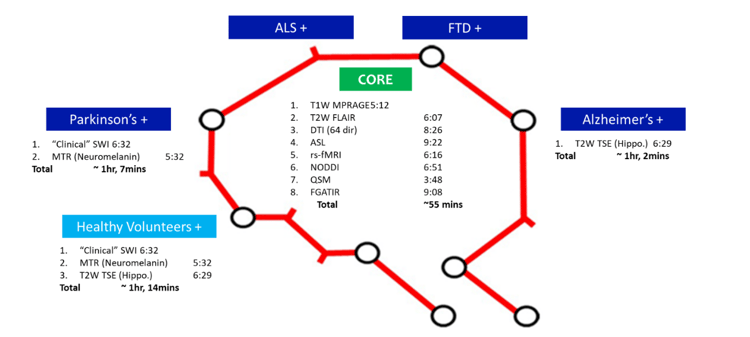
The MIND MAPS program is structured in a hub-and-spokes design with Invicro’s London location being the central locus for all imaging activities and individual studies performed by associated academic groups in order to standardize protocols.Imaging procedures include PET imaging with [18F]BCPP-EF, [11C]SA-4503 and [11C]UCB-J, to quantify the density of the MC1, s1R and SV2A respectively. All subjects also undergo a multi-modal MRI examination and clinical and cognitive evaluation.The group of healthy volunteers serve as a comparison group for all the patient cohorts.
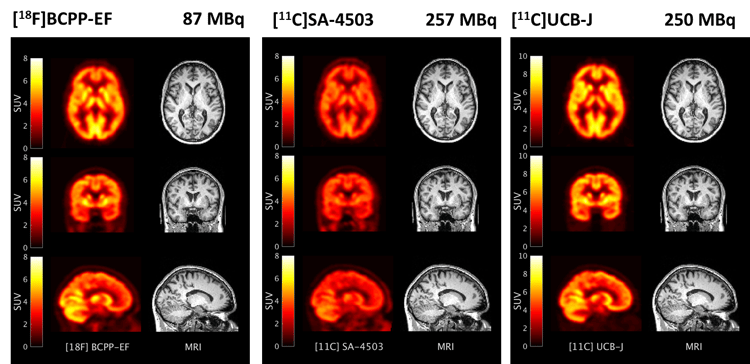
The pathophysiology of neurodegenerative disorders is multifaceted and involves many processes (see figure #1). The sequence of these processes is not clearly established, but cellular stress, involving mitochondrial and endoplasmic reticulum (ER) dysfunction is a finding common to all neurodegenerative diseases. Progressive mitochondrial dysfunction leads to energy deficits, particularly in areas of high metabolic demand such as the synapse and exacerbates protein misfolding in the ER (a notoriously energy-demanding process) further exacerbating cellular stress via UPR. Synaptic dysfunction results in neurotransmitter dysregulation, synaptic loss and axonal dieback leading to neuronal loss. We believe that the mitochondrial/ER/synapse axis will demonstrate dysfunction early in the disease and presents an attractive target system to monitor the progression of neurodegenerative disorders. Radioligands [18F]BCPP-EF, [11C]SA-4503 and [11C]UCB-J have been shown to be suitable for investigating the density of the mitochondrial complex I (MC1), the sigma 1 receptor (s1R) and the synaptic vesicular protein type 2A (SV2A) respectively, as molecular markers of the mitochondrial/ER/synaptic axis.
Completed - Details coming soon!

Completed - Details coming soon!

Completed - Details coming soon!

Ongoing - Check back at later date for completion of project.

Ongoing - Check back at later date for completion of project.

Funded/Set-up - Check back at later date for completion of project.
Under discussion/funding - Check back at later date for start of additional projects.
Under discussion/funding - Check back at later date for start of additional projects.

Under discussion/funding - Check back at later date for start of additional projects.